Solution in Chemistry
What is a Solution?
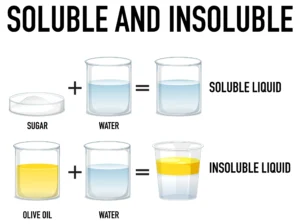
A homogeneous mixture of two or more substances is called a solution.

A solution consists of two components.
- Solute
- Solvent
Solution = Solute + Solvent
Solute → The component present in a smaller quantity within the solution is called the solute.
Solvent → The component present in a larger quantity within the solution is called the solvent.
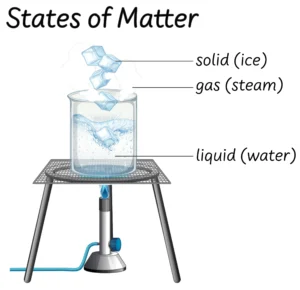
Types of Solutions — Based on the physical state of the solvent, solutions are classified into three types.
Types of Solutions
| S.No. | Types of Solutions | Solute | Solvent | Example |
| 1. | Gaseous Solutions | Gas | Gas | N₂ + O₂ Gas |
| Liquid | Gas | Chloroform + N₂ | ||
| Solid | Gas | Camphor + N₂ gas | ||
| 2. | Liquid Solutions | Gas | Liquid | Water + O₂ |
| Liquid | Liquid | Ethanol + Water | ||
| Solid | Liquid | Sugar + Water | ||
| 3. | Solid Solutions | Gas | Solid | H₂ + Pd |
| Liquid | Solid | Hg + Na | ||
| Solid | Solid | Copper + Gold (Cu + Au) |
Example — Sugar + Water