Osmosis and Osmotic Pressure.
The spontaneous flow of solvent molecules from a less concentrated solution to a more concentrated solution through a semi-permeable membrane is called osmosis.
- Solution – High Concentration Solution – Low Concentration
- Solvent – Low Concentration Solvent – High Concentration
Osmotic Pressure.
The additional external pressure applied to a solution that prevents osmosis is called osmotic pressure.
Or
The additional external pressure applied to a solution that prevents the flow of solvent molecules from a lower concentration solution to a higher concentration solution.
Due to osmosis of the solvent, the level of the solution in the Thessel funnel increases.
Note– This flow continues until equilibrium is established between the two solutions.
Membranes that allow solvent molecules to pass through but not solute molecules are called semi-permeable membranes.
The pressure produced by osmosis is called osmotic pressure.
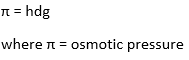
h = height of the column of solution
d = density of the solution
g = gravitational acceleration
To prevent osmosis, an additional pressure equal to the osmotic pressure must be applied to the solution.
Description.
1. First, take a rectangular vessel, separated into two equal parts by a semi-permeable membrane (SPM).
2. Pistons P1 and P2 are installed in both vessels. Fill both parts, A and B, with pure solvent and solution, respectively.
3. Due to osmosis, the molecules of the pure solvent move towards the solution. This causes piston P2 to rise upwards and P1 to move downwards.
4. Now, external pressure is applied to bring piston P2 back to its initial state and establish equilibrium. This external pressure is necessary to prevent osmosis. That is, the additional external pressure applied on the piston due to which the solvent molecules are unable to pass through the semi-permeable membrane is called osmotic pressure.
The osmotic pressure of a solution depends on the following factors.
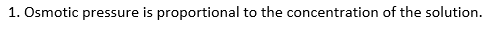

Note– Osmotic pressure is a colligative property used to determine the molar mass of large molecules, polymers, etc.
The reasons for this are.
1. Osmotic pressure can be measured at room temperature.
2. The magnitude of osmotic pressure is larger than other colligative properties.
3. In osmotic pressure, concentration is expressed in molality (m) instead of molarity (M).
Examples of osmosis.
1. Wilted flowers and carrots swell when placed in water.
2. Raw mangoes shrink when soaked in salt water for pickling.
3. Blood cells shrink when kept in salt water.
Q. What happens when raw mangoes are placed in a concentrated saline solution?
Ans. When placed in a concentrated saline solution, they expel water due to external osmosis and shrink.
Types of solutions based on osmotic pressure – Three types
1. Isotonic solution
2. Hypotonic solution
3. Hypertonic solution
1. Isotonic solution.
Two solutions whose concentration and osmotic pressure are the same at a given temperature are called isotonic solutions.
Or
Two solutions with the same osmotic pressure at a given temperature are called isotonic solutions.
When such solutions are separated by a semi-permeable membrane, osmosis does not occur between them.
Example- The solution present in red blood cells has a concentration of 0.9% (W/V) NaCl, which is an isotonic solution.
2. Hypotonic Solution.
If the osmotic pressure of two solutions is unequal, the solution with the lower osmotic pressure is called a hypotonic solution.
Example- If the salt concentration is less than 0.9%, (W/V) water will flow into the cell, causing it to swell.
3. Hypertonic Solution
If the osmotic pressure of two solutions is unequal, the solution with the higher osmotic pressure is called a hypertonic solution.
Example- If blood cells are placed in a NaCl solution greater than 0.9%, (W/V) water will flow out of the cell, causing it to shrink.
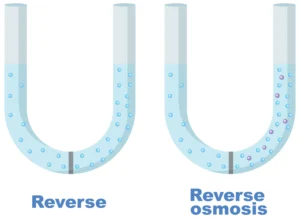
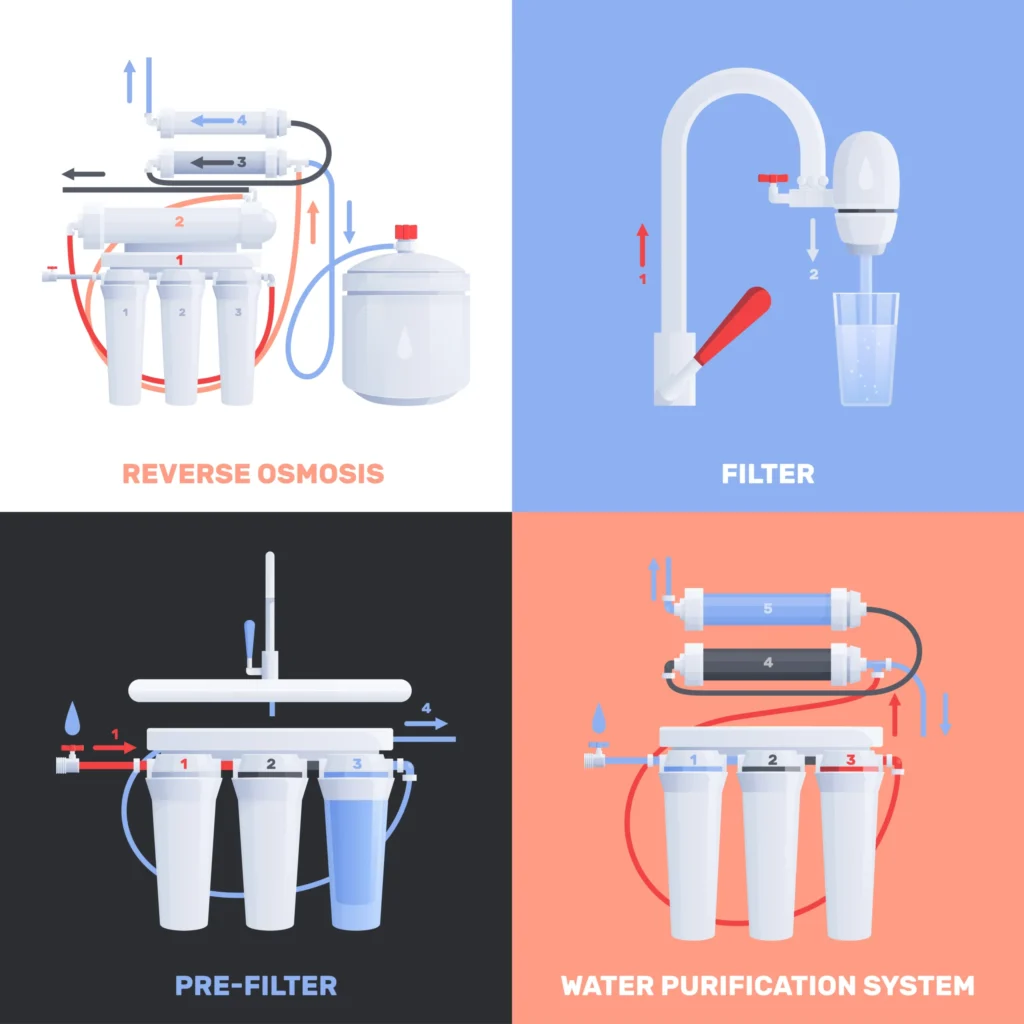
Reverse Osmosis.
When a solution is subjected to pressure greater than the osmotic pressure, reverse osmosis occurs.
Reverse Osmosis – When external pressure greater than the osmotic pressure is applied to a solution, the osmotic process occurs in the opposite direction, meaning that solvent molecules move from a more concentrated solution to a less concentrated solvent through a semi-permeable membrane. This process is called reverse osmosis (RO).
Applications.
1. Reverse osmosis is used in the desalination of seawater.
2. Water is made potable using an RO device.
Note – The semi-permeable membrane in reverse osmosis is made of cellulose acetate.
Q. Name the semi-permeable membrane most commonly used in the laboratory. How is it made?
A. This is copper ferrocyanide Cu2 [Fe(CN)6]. It is prepared by mixing equimolar aqueous solutions of CuSO4 and K4 [Fe(CN)6].