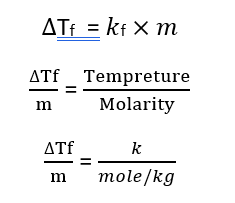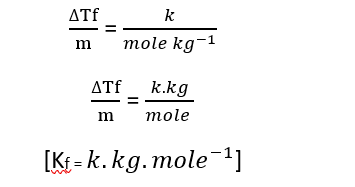Colligative Properties and the Calculation of Molecular Mass.
Properties that depend on the quantity (or the number of molecules) of the solute present in a solution—rather than on the nature of the solute itself—are known as colligative properties.
- Colligative properties are of four types.
- Relative lowering of vapor pressure
- Elevation in boiling point
- Depression in freezing point
- Osmotic pressure
1. Relative Lowering of Vapor Pressure– When a non-volatile solute is added to a pure solvent, the vapor pressure of the resulting solution is always lower than that of the pure solvent. This phenomenon is termed the *lowering of vapor pressure*. In a solution containing a non-volatile solute, the total vapor pressure is attributed solely to the solvent.
Let P1 be the vapor pressure and X1 be the mole fraction of the solvent in the solution, and let P1° be the vapor pressure of the pure solvent. Therefore, according to Raoult’s Law.

Lowering of Vapor Pressure.
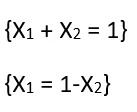
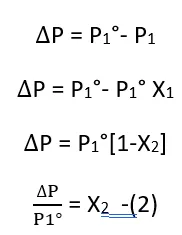
The relative lowering of vapor pressure of a solution is equal to the mole fraction of the solute present in the solution.
From Equation (2)
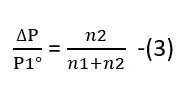
Let us assume that the mass of the solvent is W1 and its molar mass is M1, and the mass of the solute is W2 and its molar mass is M2. Then.
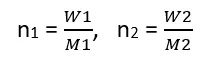
Substituting these values into Equation (3)
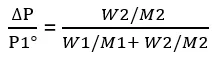
For very dilute solutions, W1/M1 >>> W2/M2. Therefore, by treating W2/M2 as negligible.
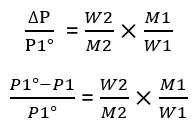
2. Elevation in Boiling Point.
Boiling Point– The boiling point of a liquid is the temperature at which the vapor pressure of that liquid becomes equal to the atmospheric pressure.
Elevation in Boiling Point– When a solid solute is added to a pure solvent, the boiling point of the resulting solution increases compared to the boiling point of the pure solvent.
OR
If a certain amount of a non-volatile solute is added to a solvent, the boiling point of the solution becomes higher than that of the solvent.
Explanation – The vapor pressure of a solution is lower compared to that of the pure solvent. To raise the vapor pressure of the solution to equal the atmospheric pressure, it must be heated to a higher temperature than the solvent; therefore, the boiling point of the solution is higher than that of the pure solvent.
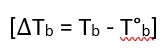

Based on experimental observations, it has been concluded that for dilute solutions, the elevation in boiling point is directly proportional to the molality (m) of the solution.
That is.
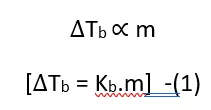
Where Kb = Boiling point elevation constant.
Let us assume that the mass of the solvent is W1 g, the mass of the solute is W2 g, and its molar mass is M2; then.
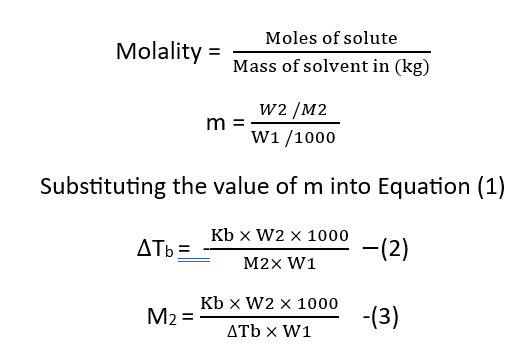
If W2 grams of a solute with molar mass M2 are present in W1 grams of a solvent, then the molality of the solution is given by the above equations.
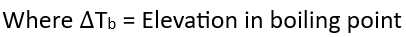
M2 = Molar mass
3. Depression in Freezing Point
Freezing Point– The freezing point of a liquid is the temperature at which the vapor pressure of the liquid phase becomes equal to the vapor pressure of its solid phase.
At the freezing point, the liquid and solid phases coexist in a state of mutual equilibrium.
Depression in Freezing Point– When a solid solute is added to a pure solvent, the freezing point of the resulting solution is lower than that of the pure solvent; this phenomenon is known as the depression in freezing point.
OR
If a certain amount of a non-volatile solute is added to a solvent, the freezing point of the solution decreases relative to the freezing point of the pure solvent. This depression in freezing point is a colligative property, meaning it depends on the number of solute particles present in the solution.
Reason– The vapor pressure of a solution is lower than the vapor pressure of the pure solvent; therefore, the vapor pressure of the solution in its liquid phase becomes equal to the vapor pressure of its solid phase only at a lower temperature. Consequently, the freezing point of the solution is lower than that of the solvent.
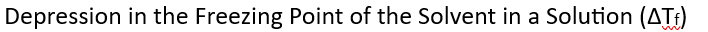
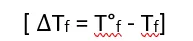
Based on experimental observations, it has been concluded that for dilute solutions, the depression in the freezing point is directly proportional to the molality of the solution.
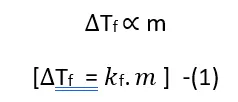
Let us assume that the mass of the solvent is W1 grams, the mass of the solute is W2 grams, and its molar mass is M; then-

Units of the Boiling Point Elevation Constant or Freezing Point Depression Constant.