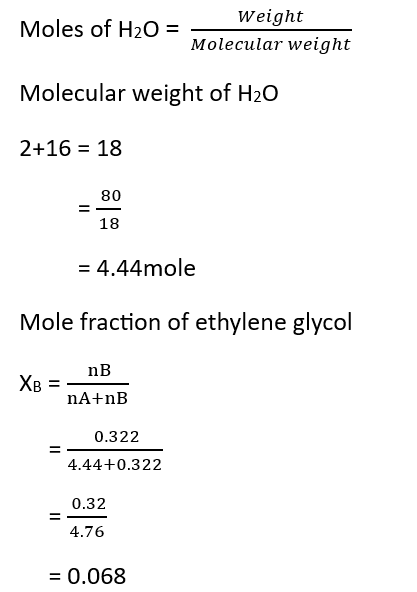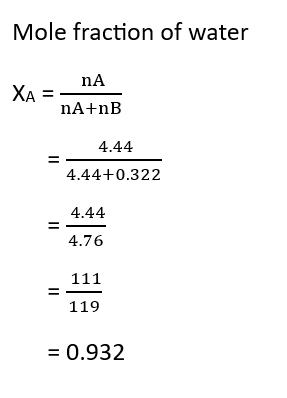Concentration of Solution.
Concentration – The amount of solute present in a unit volume of a solution is called the concentration of the solution.
The concentration of a solution is expressed in the following terms.

Methods of measurement.
1. Mass percent (w/w)%
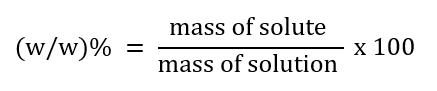
For example: 10% (w/w)% means a solution of glucose in water.
10 gm of glucose is dissolved in 90 gm of water.
2. Volume percent (v/v)%
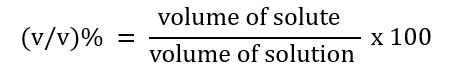
For example: 10% (v/v)% means a solution of ethanol in water.
10 ml of ethanol is dissolved in 90 ml of water.
3. Mass-Volume Percentage (W/V) %
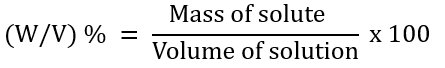
For example: Pharmacy (in medicines)
4. Parts per million (PPM). This term of concentration is used when the amount of solute is very small.
For example: 1. Dissolved oxygen in water
2. Amount of soluble salts in drinking water
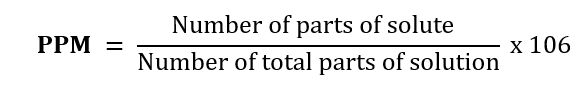
5. Mole Fraction – The mole fraction of a component/component in a solution is equal to the ratio of the number of moles of that component to the total number of moles present in the solution.
Suppose the number of moles of two components A and B in a binary solution are nA and nB respectively, and the mole fractions of these components are xA and xB respectively. Therefore,
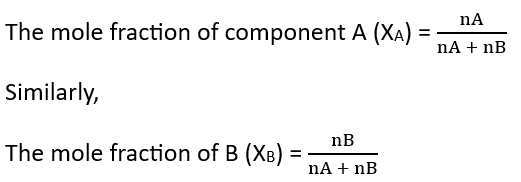
Note: The sum of the mole fractions of all components in a solution is 1.
6. Molarity – The number of moles of solute present in 1L of a solution is called molarity.
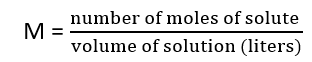
Question: A solution is prepared by dissolving 10g of NaOH in 250ml of water. Find the molarity.
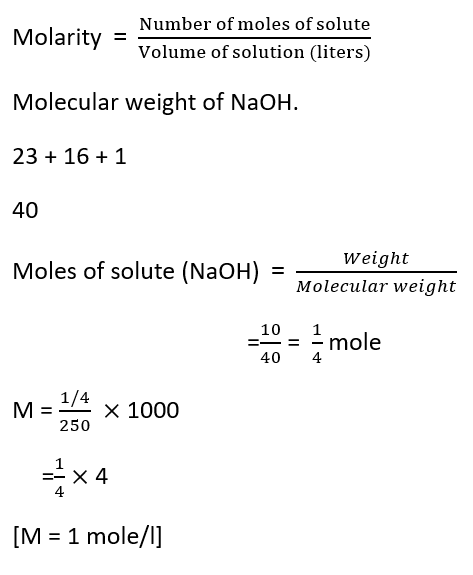
Question: Calculate the molarity of a solution containing 5g of NaOH dissolved in 450ml of solution.
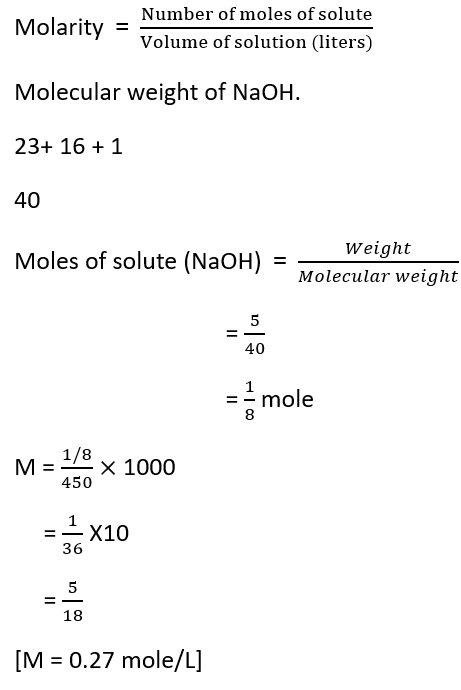
7. Molality (m) – The number of moles of solute present in 1kg of solvent is called molality. It is denoted by m.
Or
The number of moles of solute dissolved in 1kg of solvent is called molality.
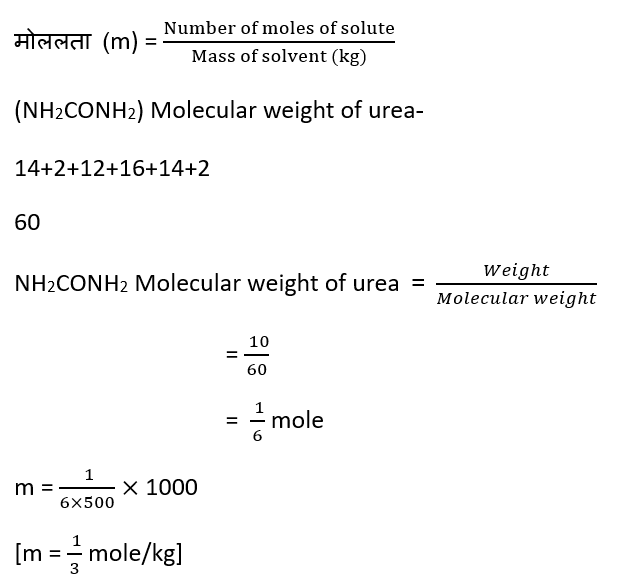
Question – A solution is prepared by dissolving 10g of urea (NH2CONH2) in 500g of water. Find the molality of the solution.
Question – 0.05 mole of ethanoic acid is dissolved in 250 grams of benzene. Find the molality of the solution.
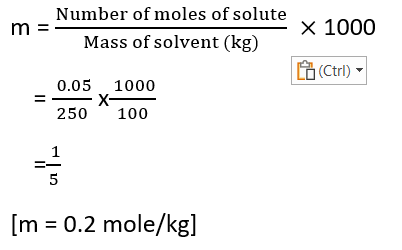
Question – Which of the following, molality or molarity, represents the concentration of a solution more accurately? Why?
Answer: Molality represents concentration more accurately because it is not affected by temperature.
Note: Mass percent, mole fraction, ppm, and molality (m) do not depend on temperature.
While molarity (M), mass-volume percent, and volume percent depend on temperature.
Question – Unit of molarity?
Answer: Mole/L
Question – A 35% (V/V) solution of ethylene glycol is used to cool vehicle engines. Find the volume of water in ml.
Answer: Ethylene glycol is 35% (V/V). Therefore, if the total volume is 100 ml, the volume of water will be
100 – 35 = 65 ml.
Therefore, the volume of water in this solution will be 65ml or 65%.
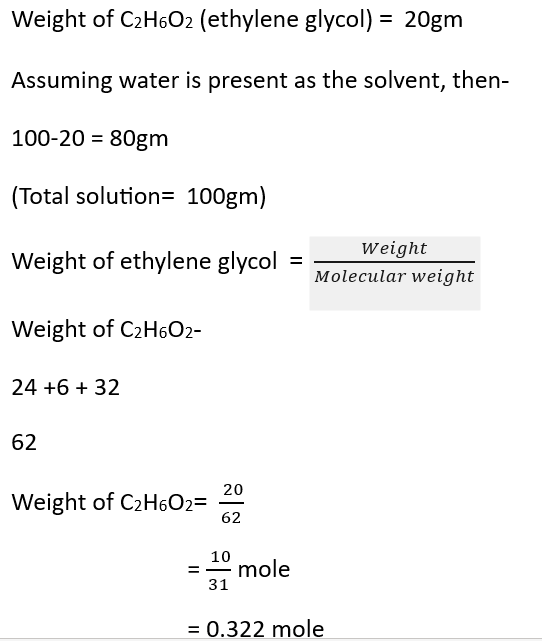
Question – Calculate the mole fraction of ethylene glycol (C2H6O2) if 20% of the mass of C2H6O2 is present in the solution.