Solubility – At a specific temperature, the maximum amount of solute present in a unit volume of a solution is termed the solubility of that substance.
Saturated Solution – “A solution in which no further solute can be dissolved at a given temperature and pressure is called a saturated solution.”
Unsaturated solution – A solution in which additional solute can still be dissolved at the same temperature is called an unsaturated solution.
Solubility of Solids in Liquids.
1. Nature of the Solute and Solvent – Polar solutes dissolve in polar solvents, whereas non-polar solutes dissolve in non-polar solvents; that is, “like dissolves like.”
Example – NaCl (common salt) and sugar dissolve easily in water, whereas naphthalene and anthracene do not.
2. Temperature – The effect of temperature on the solubility of a solid in a liquid depends on whether the process of dissolution (solvation) is endothermic or exothermic.
- In an Endothermic Process – In an endothermic reaction, increasing the temperature leads to an increase in the solubility of the solid in the liquid.
- In an Exothermic Process – In an exothermic reaction, increasing the temperature leads to a decrease in the solubility of the solid in the liquid.
Example – Dissolving NaCl or glucose in water is an endothermic process; therefore, solubility increases as the temperature rises.
Conversely, dissolving NaOH in water is an exothermic process; consequently, the solubility of NaOH decreases as the temperature rises.
3. Effect of Pressure – Pressure has no effect on the solubility of solids in liquids because solids and liquids are virtually incompressible. Therefore, increasing or decreasing the pressure results in no significant change in their volume.
Factors Affecting the Solubility of Gases in Liquids.
1. Nature of the Gas – Gases that ionize in a liquid (solvent) or undergo a chemical reaction with the solvent exhibit higher solubility.
Example – HCl, NH3 , SO2 , SO3, CO2, etc., have high solubility in water.
Conversely, gases that neither ionize in the solvent nor undergo a chemical reaction with it exhibit lower solubility.
Example – H2, N2, etc.
2. Effect of Temperature – The solubility of gases in liquids decreases as the temperature increases. Increasing the temperature increases the kinetic energy of the gas molecules, causing the gas particles to escape from the liquid. The dissolution of gases in liquids is an exothermic process. According to Le Chatelier’s Principle, increasing the temperature leads to a decrease in the solubility of gases in liquids.
Exceptions – The solubility of gases such as H2 and He increases as the temperature rises.
3. Effect of Pressure – The solubility of gases in liquids increases as the pressure increases.
Henry established a quantitative relationship between the solubility of gases in liquids and pressure, which is known as Henry’s Law.
Henry’s Law.
This law states, “The solubility of a gas in a liquid is proportional to its partial pressure.”
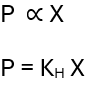
where KH = Henry’s constant
x = mole fraction of the gas
If the solubility of a gas in a liquid is expressed in mole fractions, then Henry’s law is expressed as above.
The value of Henry’s constant varies at different pressures. This means that Henry’s constant depends on the nature of the gas. As the value of Henry’s constant increases, the solubility of gases in liquids decreases.
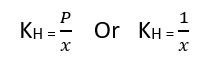
Applications of Henry’s Law.
1. CO2 gas is injected at high pressure into soda water and other soft drinks.
2. Atmospheric pressure is low in high altitudes, reducing the solubility of O2 in the blood. Consequently, the supply of O2 to the brain decreases. This condition causes a person to experience dizziness. This condition is called anoxia.
3. Divers use cylinders when diving. As the water deepens, the air pressure increases, increasing the solubility of gases. However, when a diver comes to the surface, the solubility of gases decreases due to the decrease in air pressure, causing dissolved in blood gases like N2 to escape in the form of bubbles. These N2 bubbles gradually block the arteries, causing a condition called bends. To avoid this situation, He gas is added to O2 cylinders. Getting into the bends is a life-threatening situation.
Note– The diver’s breathing tanks/cylinders are filled with air diluted with He. [11.7% He, 56.2% N2, 32.1% O2]
Limitations of Henry’s Law – Henry’s Law applies only when.
- The pressure is not high.
- The temperature is not low.
- The gas does not react with the solvent, and its ionization and association do not occur.
- The solubility of the gas is low.
Q. Are aquatic animals more comfortable in cold water than in warm water? Why?
Ans. As the temperature of water increases, the value of Henry’s constant (KH) increases for low O2 gas, which reduces the solubility of O2 in water or makes breathing difficult for aquatic animals.
Note – According to Henry’s Law, if a graph is drawn between the mole fraction of a gas and its partial pressure, a straight line is obtained, and the slope of this graph is equal to Henry’s constant.